|
Cells are extremely complex structures, and understanding the formation of abnormal structures in a disease state requires the consideration of numerous cellular mechanisms.
In general, the word "vacuole" is a term that has been used to describe voids or pockets inside a cell. Vacuoles, in some cases, are the remains of vesicles (functional vacuoles). These terms have been used interchangeably in the literature, but have special meaning depending on the context of use. Where functional roles of vesicles have been described, the same structures have also been referred to as organelles (miniature organs).
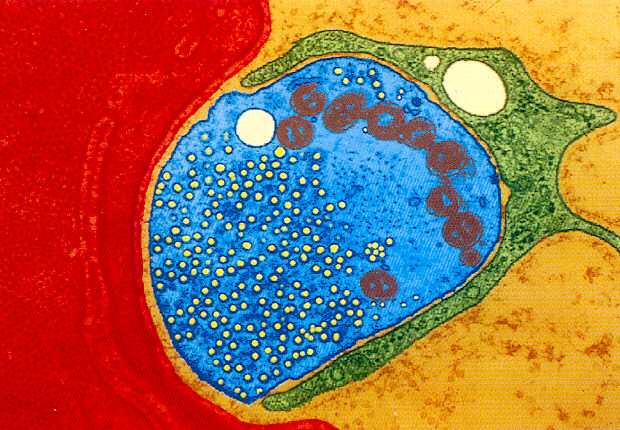
In healthy cells, especially in secretory cells (similar to that illustrated in the motor neuron above), one can observe spherical structures, or vacuoles, called condensing vacuoles. The condensing vacuoles are filled with varying amounts of secretory products. These condensing vacuoles are the transport containers which carry the synthesized products from another organelle called the Golgi complex of the cell.
In the blue portion of the illustration above, what appears to be a mass of small bubbles are actually synaptic vesicles in a motor neuron's synaptic foot (the part of the nerve that comes in close proximity to the muscle cell). These synaptic vesicles are filled with acetylcholine. The release of acetylcholine from these vesicles initiates the action potential of the muscle cell, enabling muscle contraction.
The term vacuole is also used to describe abnormal voids in muscle tissue cells. This is called vacuolization. Vacuolization is a characteristic of various neuormuscular diseases. In this case they are also referred to as inclusions. Sometimes the term inclusion is used to refer to the content of a vacuole.
To see more illustrations of vacuoles go to Vacuoles(or Inclusions) at the NDC.
The formation of vacuoles in a disease state is often associated with the activities of lysosomes and peroxisomes.
A vacuole formed by a lysosome or peroxisome can contain a wide variety of materials (the residue of enzymatic digestive activity), and may include crystallized proteins, microtubules, various other cellular debries, or watery fluid.
A lysosome is a special type of vesicle closely associated with the normal cellular process of endocytosis and intracellular synthesis and transport.
A lysosome is a pouch enclosed by a single, thin, three-layered membrane containing hydrolytic digestive enzymes. These enzymes, are used to digest (or hydrolyze) various macromolecules including: fats, proteins, polysaccharides, and nucleic acids.
Lysosomes are sometimes thought of as the clean-up crew of the cell because of the crucial role they play in phagocytosis. In this process, a cell engulfs another organism or macromolecule, creating a food vacuole. The lysosome then fuses to the vacuole and the enzymes of the lysosome digest the contents of the vacuole. As scavengers, lysosomes are active not only during cell injury or death, but also participate in the digestion of cell material derived from the programmed destruction of other cells, or even the digestion of other organelles within the same cell.
Lysosomes vary greatly in appearance and content from cell type to cell type and also in relation to the physiological state of any particular cell.
In order for lysosomes to function properly, an acidic environment is needed. Lysosomes maintain an internal acidic environment by pumping hydrogen ions into their internal cavity. A disruption of the pH balance in the cell can lead to irregularities in lysosomal activity.
There is a growing list of activities associated with lysosomes, but the one common feature in all of these is the sequestering of digestive enzymes inside the membrane where they are not disruptive to other cell functions. This protective role of the lysosome is obviously curtailed when the membrane is damaged.
Peroxisomes are similar to lysosomes in that they are compartments of enzymes enclosed in a single membrane. They differ from Lysosomes in that some enzymes within peroxisomes break down fats into smaller molecules by moving a hydrogen molecule from the fat to an oxygen molecule, forming hydrogen peroxide. These fats are broken down for transportation to the mitochondria (the cells energy factory). These broken down fats serve as fuel for cellular respiration.
One of the primary functions of peroxisomes is to break down the hydrogen peroxide that is generated through other mechanisms during cellular metabolism.
Special enzymes in peroxisomes break down hydrogen peroxide into water and molecular oxygen. Peroxisomes are also used to detoxify poisons like alcohol through similar enzymatic processes.
These vacuoles differ from lysosomes in that they contain the enzyme catalase. This suggests that part of their function is to protect the cell from the damaging effects of the hydrogen peroxide generated during metabolism.
It is also known that most cells can take up materials from their immediate environment by the process of endocytosis. This can be described as the cell membrane forming a pocket that closes around a bit of the extracellular fluid. This pocket forms a vacuole or vesicle called an endocytotic vesicle. Others, containing only the liquid part of the extracellular fluid are called pinocytotic vesicles, and the process is called pinocytosis.
Similar processes are associated with the transport of cholesterol and other lipid components. In general, these processes are closely involved in the bulk transport of materials into and out of the cell and various cell structures, and are closely associated with cell metabolism.
While these functional vacuoles apparently help manage the availability and transport of materials involved in cellular structure, membrane integrity, and energy production, they also use significant amounts of cellular energy to carry out their activities, and clearly posses significant destructive potential if their own integrity has been violated.
In striated muscle, the formation of muscle fiber and other functions is dependent on proper pH and ionic strength of the cellular environment. Changes in acidity or alkalinity can effect the solubility of the muscle proteins actin and myosin. These conditions can also influence the free energy exchange in muscle metabolism and other processes associated with the binding of ATP to myosin, a critical step in muscle function.
ATP is of key importance to muscle contraction and numerous other processes. All of these critical processes are dependent on specific enzyme activity including the work of CPK (Phosphocreatine Kinase).
Another important point to consider is the critical role of the calcium ion in muscle protein function and ATP hydrolysis. Acid-base balance (involving all the principle ions including calcium, sodium, potassium, chloride and phosphate) plays an important role in all muscle functions. Calcium control is exerted by its effect on the activity of the kinase enzyme. This effect seems to be regulated by the presence of a calcium-binding protein known as calmodulin.
It is known that the mechanisms involved in the periodic paralyses include ion metabolism. In Hypo and Hyperkalemic Periodic Paralysis the sodium and calcium membrane channels are identified as sites of dysfunction. Pharmacological control of these disease states is dependent on the manipulation of pH, ion excretion, and related mechanisms. It has also been demonstrated that in hypokalemic periodic paralysis ions are able to enter the cell but not escape. Clearly this modifies the ionic balance of the cytoplasm, influencing the behavior of lysosomes, and other enzymatic functions.
In the dystrophic syndromes, including myotonic dystrophy, inclusion body myositis, debranching enzyme deficiency, distal mitochondrial myopathy, and others, the CK enzyme level is usually normal or slightly increased. Muscle biopsy shows intranuclear tubular filaments and rimmed vacuoles in the muscle fibers.
Ultrastructural studies show that the rimmed vacuoles contain myeloid structures and other cytoplasmic degradation products and that the inclusions consist of microtubular filaments.
The differential diagnosis in these conditions, includes motor neuron disease, the distal muscular dystrophies, peripheral neuropathies, and polymyositis. The diagnosis in these conditions is usually clarified by a careful study of the muscle biopsy. Many of these conditions have an immune component associated with them. Again, here is another possible implication of the lysosomes.
There is a long and growing list of related diseases associated with the various myopathies, including the periodic paralyses.
Muscle fiber necrosis (death, deterioration) and vacuolization are common in many of these disorders. While in some cases, vacuolization can possibly be reversed if the contributing conditions are eliminated, one would certainly be concerned about those channelopathies that result in a modification of the pH and ionic balance of the cell, as this could lead to a chronic disturbance of lysosomal activity.
Note: The above article summarizes materials from a number of sources including: The Cecil Textbook of Medicine, 20th Edition, 1996, and Cell Structure & Function, an Integrated Approach, 3rd Edition, 1991. These notes are specifically selected for their possible relevance to understanding mechanisms of permanent muscle weakness (PMW) and to the vacuolization that is sometimes found in cases of periodic paralysis.
|